Last month, Pax Labs, Inc. introduced a new e-cigarette technology: the use of nicotine salts rather than free-base nicotine as a core ingredient of the e-liquid. The new product, called the JUUL, provides a blood nicotine absorption profile which is similar to that for traditional cigarettes, unlike most electronic cigarettes currently on the market.
According to an article in Tech Crunch:
"While about 60 percent of smokers have at one time or another tried e-cigs, few come back to them. The majority return to traditional cigarettes, in part because e-cigs don’t provide the same sensation as lighting up a Marlboro or other brand. For the past two years, Pax Labs has been working to change that — first by attempting to reverse engineer the chemistry behind how people’s bodies react when smoking, and then by building a vaporizing device to deploy that technology. The result is an extremely attractive and compact little device that packs the same amount of nicotine as a pack of cigarettes into a tiny liquid-nicotine cartridge. One of the key findings in the development of its liquid-nicotine “juice” was the use of nicotine salts found in leaf-based tobacco as the core ingredient. By extracting and using those salts, Pax is able to create an experience that is more like smoking than other e-cigarettes on the market. That includes offering up two times the nicotine strength and three times the vapor quality of competing products. To figure out if that was true, Pax commissioned study to look at the impact of the Juul compared with traditional combustible cigarettes and other e-cigarettes. Here’s a graph of what they found:"
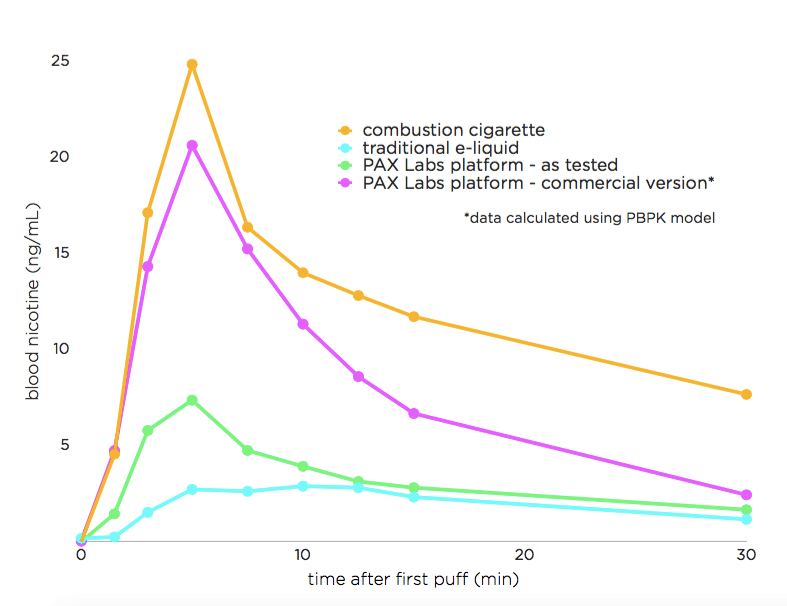
One of the reasons why some smokers do not find e-cigarettes satisfactory as an alternative is that they do not feel the nicotine "hit" that they are used to with their regular cigarettes. This is most likely because nicotine delivery in most e-cigarettes is not nearly as good as in tobacco cigarettes. In particular, the peak blood nicotine level is not nearly as high as with traditional cigarettes. Without this "spike" in blood nicotine, users may not feel a nicotine "hit." The JUUL addresses this problem by using nicotine in its salt form, leading to a more dramatic spike in peak blood nicotine levels.
New Methods to Regulate E-Cigarette Temperature
While the media have recently spread misinformation about "scary" levels of formaldehyde produced by some e-cigarettes, there have been products on the market that have overheating protection, thus avoiding the formaldehyde problem completely. By controlling heater temperature and output wattage, it is possible to carefully control the heating profile of the e-liquid, thus optimizing vapor production while eliminating the problem of formaldehyde formation. This explains why testing of products such as Vuse revealed no detectable level of formaldehyde.
There are companies which develop hardware that can be used by e-cigarette manufacturers to prevent overheating. One such company is Evolv Inc., whose hardware allows both open and closed vapor delivery systems to enjoy temperature regulation and overheating protection. Thus, it is now possible for users to be able to control the voltage applied to their personal vaporizers, while at the same time not taking a risk of overheating the e-liquid and being exposed to degradation products such as formaldehyde and acrolein. In other words, it is not necessary for the FDA to ban all open systems. Instead, it can simply require temperature regulation and overheating protection.
The Rest of the Story
These examples of major innovations in e-cigarette and personal vapor delivery systems reveal the folly of the FDA's proposed electronic cigarette regulatory scheme. The centerpiece of that scheme is the requirement for all new and existing products to submit new product applications. The net effect of such a regulation would be to stifle innovation. Such a regulation would put a large and unnecessary obstacle in the way of the development, introduction, and marketing of new, innovative, more effective, and safer products.
The electronic cigarette space is perhaps one of the most rapidly evolving of any consumer product. The newer products on the market are almost uniformly more effective and safer than the older products. It therefore makes absolutely no sense to require these new products to be required to obtain pre-approval. If anything, it is the older products - which do not have safety protection characteristics that are readily available - that should have to justify their presence on the market.
My view is that there is no justification for any requirement for product pre-approval, either for existing or new electronic cigarettes. Instead, the FDA should simply promulgate a set of minimum safety and quality control standards for all products. By making overheating protection part of these standards, the agency can prevent the formaldehyde and acrolein problem and put an end to the hysteria.
One of the major flaws of the arguments being made by e-cigarette opponents is that these arguments are largely based on antiquated systems. For example, the FDA is still talking about the diethylene glycol problem, even though that problem was solved years ago. It is only a matter of time before these recent advances in technology reach all e-cigarette and vapor delivery systems. Thus, the question is not whether electronic cigarettes on the market in 2007 have net public health benefits, but whether the e-cigarettes on the market in the future will have net public health benefits.
The answer is clearly yes, but the degree to which those benefits are realized depends largely on how the FDA chooses to regulate these products. If the FDA requires new product applications, it will stifle innovation and slow down the evolution of safety and effectiveness improvements. However, if the agency instead eschews the formation of a huge bureaucracy in favor of simply establishing minimum safety standards, it can help maximize the benefits of electronic cigarettes in protecting the public's health by optimizing the ability of these alternative nicotine delivery products to compete successfully with traditional combusted tobacco cigarettes.
Thanks for the great introductions.
ReplyDeleteWell, that's probably good enough. I have just get my won custom juul case last time and was glad to say I was satisfied with the design. I really enjoy using the vape with coolest designs and flavors.
ReplyDeleteWell the story has been properly define and people would get the proper awareness of it with the means of it and they can easily finalize that thing and Juul Pod System Wholesale is also looking for this thing and they can find the perfectness of it.
ReplyDeleteWell, your blog says it all. I could not say more about this. I can only say you may try and make Custom Juul for creativity and relaxation.
ReplyDelete